Spearheading Safety, Clinical Efficacy & Regulatory Compliance Using Advanced Genetic Engineering & Novel Delivery Platforms to Shape the Next Era of Genome Editing
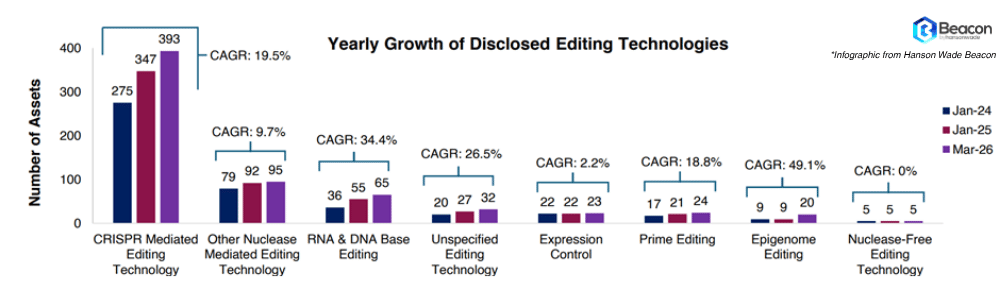
The FDA has shown its' hand. The Plausible Mechanism Framework and new NGS safety guidance signal one thing clearly: regulators want genome editing therapies to reach patients. Biopharma are also pushing back with a surge of novel genome editing assets deployed in the last 2 years, targeting rare and prevalent diseases with scarce treatment options.
But intent and support alone won't get these therapies to patients. Developmental, manufacturing, and regulatory challenges still stand between the lab and the patients waiting.
That's why the 7th Genome Editing Therapeutics Summit returns to Boston in 2026 where R&D, preclinical, translational, regulatory, and c-suite leaders will come together to answer one question: how do we maximize the application of genome editing therapies to patients who need them most?
Don't miss out on insights from AbbVie, Bristol Myers Squibb, Intellia Therapeutics, Beam Therapeutics, Sanofi, and others on how genome editing can move from a promising therapy to an essential one for patients facing the most genetically complex diseases.
Attending Companies Include



































